Advertisements
Advertisements
प्रश्न
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
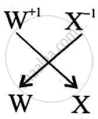
What is the formula of the compound formed between W and X.
Advertisements
उत्तर
WX is the formula of the compound formed between W and X.
APPEARS IN
संबंधित प्रश्न
State differences between the properties of ionic compounds and covalent compounds.
Fill in the blanks.
The electrovalent bond or ionic bond is called as _____ bond
How many atoms of each kind are present in the following molecule:
Calcium oxide
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one :
State type of bond between Y and Z and give its molecular formula.
State the following is a reduction reaction or oxidation reaction.
\[\ce{A^3+ + e- -> A^2+}\]
Name an element which does not contain neutron.
What do you understand by redox reactions?
State the type of bonding in the following molecules.
Hydrogen chloride
Explain Octet rule with an example.
How many atoms of each kind are present in the following molecule:
water
