Advertisements
Advertisements
प्रश्न
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
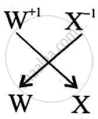
What is the formula of the compound formed between W and X.
Advertisements
उत्तर
WX is the formula of the compound formed between W and X.
APPEARS IN
संबंधित प्रश्न
Fill in the blank.
___________ single covalent bonds are formed between the carbon and chlorine atoms.
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms a cation?
Why do they exist as monoatoms in molecules?
Define the term:
orbit
Draw orbit structure diagram of calcium oxide (CaO).
Explain Octet rule with an example.
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Choose the odd one out and write the reason:
The one which is composed of all the three kinds of bond.[ionic, covalent and coordinate bond]
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
