Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
Advertisements
उत्तर
Geometric atomic structure of:
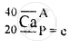
A = P + n
40 = 20 + n
∴ n = 40 − 20 = 20 neutrons
e = 20
Electronic arrangement 2, 8, 8, 2
K L M N

APPEARS IN
संबंधित प्रश्न
How do atoms attain noble gas configuration?
Define a chemical bond.
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
Give electron dot diagram of the following:
methane
How many atoms of each kind are present in the following molecule:
Calcium oxide
State the following is a reduction reaction or oxidation reaction.
\[\ce{A^3+ + e- -> A^2+}\]
Why do `""_17^35"CI"` and `""_17^37"CI"` have the same chemical properties? In what respect do these atoms differ?
How do atoms attain noble gas configurations?
Identify the following reaction as either oxidation or reduction:
K - e- → K+
How do atoms attain Noble gas electronic configuration?
