Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^39_19K}\]
Advertisements
उत्तर
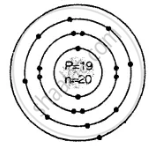
Geometric atomic structure of:
\[\ce{^39_19K}\]
Number of protons P = 19 = e
A = P + n
39 = 19 + n
∴ n = 39 − 19 = 20
e = 19 (2, 8, 8, 1)
Electronic configuration
2, 8, 8, 1 electronic arrangement
APPEARS IN
संबंधित प्रश्न
A Compound 'X' consists of only molecules. Hence 'X' will have
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
State the following is a reduction reaction or oxidation reaction.
\[\ce{Pb -> Pb^2+ + 2e-}\]
State one observation which shows that atom is not indivisible.
How does the Modern atomic theory contradict and correlate with Dalton's atomic theory?
Give the orbital diagram of the following:
Magnesium chloride
The valency of noble gases is ______.
Match the following:
| 1. | Atomic bond | a. | Oxygen and hydrogen |
| 2. | Atoms with different electronegativities | b. | acceptor bond |
| 3. | An atom that accepts electron pair | c. | covalent bond |
| 4. | Rusting of iron | d. | donor atom |
| 5. | An atom that provides electron pair | e. | oxidation |
| f. | ionic bond |
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
