Advertisements
Advertisements
Question
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^39_19K}\]
Advertisements
Solution
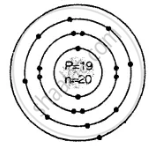
Geometric atomic structure of:
\[\ce{^39_19K}\]
Number of protons P = 19 = e
A = P + n
39 = 19 + n
∴ n = 39 − 19 = 20
e = 19 (2, 8, 8, 1)
Electronic configuration
2, 8, 8, 1 electronic arrangement
APPEARS IN
RELATED QUESTIONS
Fill in the blanks.
The electrovalent bond or ionic bond is called as _____ bond
Match the column A with Column B.
|
Column A |
Column B |
|
(i) Sodium chloride (ii) Ammonium ion (iii) Carbon tetrachloride |
Covalent bond Ionic bond Covalent and coordinate bond |
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms a cation?
Explain
Duplet rule for the formation of hydrogen
Draw orbit structure diagram of sodium chloride (NaCl).
State the type of bonding in the following molecules.
Calcium oxide
Copy and complete the following table relating to the atomic structure of some elements:
| Element | Atomic Number | Mass Number | Number of protons | Number of Neutrons | Number of Electrons |
| Beryllium | 4 | 9 | |||
| Fluorine | 9 | 10 | |||
| Sodium | 12 | 11 | |||
| Aluminium | 27 | 13 | |||
| Phosphorus | 31 | 15 |
Draw the geometric atomic structure of the three isotopes of hydrogen and the two isotopes of chlorine.
Why are Noble gases inert in nature?
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
