Advertisements
Advertisements
Question
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
Advertisements
Solution
Geometric atomic structure of:
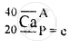
A = P + n
40 = 20 + n
∴ n = 40 − 20 = 20 neutrons
e = 20
Electronic arrangement 2, 8, 8, 2
K L M N

APPEARS IN
RELATED QUESTIONS
In term of electron transfer, define
Reduction
Fill in the blank.
___________ single covalent bonds are formed between the carbon and chlorine atoms.
State the following is a reduction reaction or oxidation reaction.
\[\ce{Pb -> Pb^2+ + 2e-}\]
How are X-rays produced?
Complete the following table relating to the atomic structure of some elements.
|
Element Symbol |
Atomic Number |
Mass Number |
Numbers of neutrons |
Number of Electrons |
Number of Protons |
|
Li |
3 |
6 |
|
|
|
|
Cl |
17 |
|
20 |
|
|
|
Na |
|
|
12 |
|
11 |
|
Al |
|
27 |
|
|
13 |
|
S |
|
32 |
16 |
|
|
Draw orbit structure diagram of calcium oxide (CaO).
Give the orbital diagram of the following:
Magnesium chloride
State the type of bonding in the following molecules.
Hydrogen chloride
Choose the odd one out and write the reason:
Complete the following table:
| a. | Dativebond | ______ |
| b. | CaH2 → Ca+H2 | ______ |
| c. | ______ | Reduction |
| d. | ______ | Redox reaction |
