Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
Advertisements
उत्तर
Geometric atomic structure of:
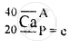
A = P + n
40 = 20 + n
∴ n = 40 − 20 = 20 neutrons
e = 20
Electronic arrangement 2, 8, 8, 2
K L M N

APPEARS IN
संबंधित प्रश्न
Complete the statement by filling the gaps using appropriate term from the terms given in the bracket.
(slow, coloured, arrow, fast, smell, milky, physical, product, chemical, reactant, covalent, ionic, octet, duplet, exchange, sharing, equality sign)
Sodium chloride is ______ compound while hydrogen chloride is ______ compound.
State differences between the properties of ionic compounds and covalent compounds.
Choose the correct option
Which of the following is the correct electronic configuration of potassium?
Explain
Duplet rule for the formation of hydrogen
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of reduction.
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidizing agent.
Give the orbital diagram of the following:
Magnesium chloride
Identify the following reaction as either oxidation or reduction:
O + 2e- → O2-
How many atoms of each kind are present in the following molecule:
water
Elements A, B and C have atomic number 17, 19 and 10 respectively.
Write down the formula of the compound formed by two of the above elements.
