Advertisements
Advertisements
प्रश्न
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
Advertisements
उत्तर
(a) i.
|
|
\[\ce{^24_12Mg}\] |
\[\ce{^26_12Mg}\] |
|
|
|
|
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, the composition of nuclei
\[\ce{_12Mg^24->(P,n),(12+12)_12Mg^26->(p,n)(12+14)}\]
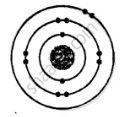
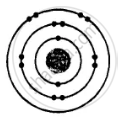
ii. Electronic configuration = 2, 8, 2
(b) Mass numbers of two isotopes of magnesium are different because of the different number of neutrons, i.e. 12 and 14, respectively.
संबंधित प्रश्न
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms an anion?
In term of electron transfer, define
Reduction
What type of bond is formed between two atoms, when the electronegative difference between them is low ?
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of reduction.
In the formation of magnesium chloride (by direct combination between magnesium and chlorine), name the substance that is oxidized and the substance that is reduced.
Copy and complete the following table relating to the atomic structure of some elements:
| Element | Atomic Number | Mass Number | Number of protons | Number of Neutrons | Number of Electrons |
| Beryllium | 4 | 9 | |||
| Fluorine | 9 | 10 | |||
| Sodium | 12 | 11 | |||
| Aluminium | 27 | 13 | |||
| Phosphorus | 31 | 15 |
Match the following:
| 1. | Monoatomic gaseous atom | a. | Electrovalent bond |
| 2. | Octet rule | b. | Benzene |
| 3. | Ionic bond | c. | Water |
| 4. | Non-polar solvent | d. | Electronic theory of valence |
| 5. | Polar solvent | e. | Noble gases |
How many atoms of each kind are present in the following molecule:
Chlorine
How many electrons are required or released by each atom in water molecule to attain the nearest noble gas configuration?