Advertisements
Advertisements
प्रश्न
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
Advertisements
उत्तर
(a) i.
|
|
\[\ce{^24_12Mg}\] |
\[\ce{^26_12Mg}\] |
|
|
|
|
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, the composition of nuclei
\[\ce{_12Mg^24->(P,n),(12+12)_12Mg^26->(p,n)(12+14)}\]
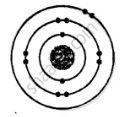
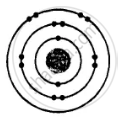
ii. Electronic configuration = 2, 8, 2
(b) Mass numbers of two isotopes of magnesium are different because of the different number of neutrons, i.e. 12 and 14, respectively.
संबंधित प्रश्न
Define the term:
orbit
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidizing agent.
Give the orbital diagram of the following:
Nitrogen
State the type of bonding in the following molecules.
Hydrogen chloride
Identify the following reaction as either oxidation or reduction:
K - e- → K+
Identify the following reaction as either oxidation or reduction:
Fe3+ + e- → Fe2+
Identify the following reaction as either oxidation or reduction:
Zn → Zn2+ + 2e-
Copy and complete the following table relating to the atomic structure of some elements:
| Element | Atomic Number | Mass Number | Number of protons | Number of Neutrons | Number of Electrons |
| Beryllium | 4 | 9 | |||
| Fluorine | 9 | 10 | |||
| Sodium | 12 | 11 | |||
| Aluminium | 27 | 13 | |||
| Phosphorus | 31 | 15 |
Choose the odd one out and write the reason:
How many atoms of each kind are present in the following molecule:
water