Advertisements
Advertisements
प्रश्न
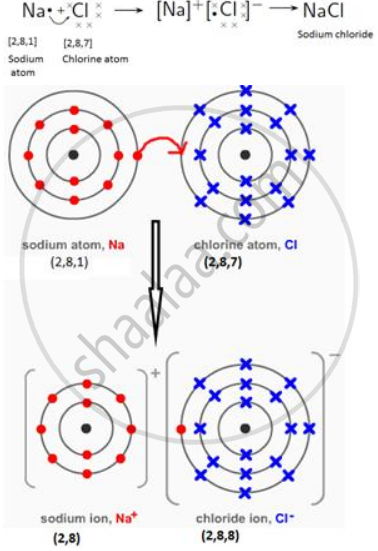
Draw orbit structure diagram of sodium chloride (NaCl).
आकृती
Advertisements
उत्तर
NaCl
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Among the following compounds identify the compound that has all three bonds (ionic, covalent and coordinate bond).
Name an element which does not contain neutron.
How does the Modern atomic theory contradict and correlate with Dalton's atomic theory?
Why do they exist as monoatoms in molecules?
Name the following:
The shell closest to the nucleus of an atom
Identify the following reaction as either oxidation or reduction:
Fe3+ + e- → Fe2+
Five atoms are labelled V to Z
| Atoms | Mass Number | Atomic Number |
| V | 40 | 20 |
| W | 19 | 9 |
| X | 7 | 3 |
| Y | 16 | 8 |
| Z | 14 | 7 |
- Which one of these atoms
(1) contains 7 protons;
(2) has an electronic configuration 2, 7? - Write down the formula of the compound formed by atoms X and Y.
Complete the following table:
| Atoms | Atomic number | Electron distribution | |
| 1. | O | 8 | ______ |
| 2. | N | ______ | ______ |
| 3. | ______ | 17 | ______ |
| 4. | ______ | ______ | 2,8,2 |
| Name of compound | Type of compound | |
| a. | Sodium chloride | ______ |
| b. | ______ | co-ordinate covalent compound |
| c. | Methane | ______ |
| d. | Fluorine molecule | ______ |
How many electrons are required or released by each atom in carbon tetrachloride to attain the nearest noble gas configuration?
