Advertisements
Advertisements
Question
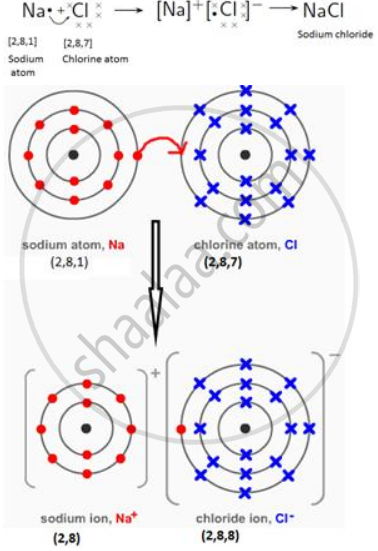
Draw orbit structure diagram of sodium chloride (NaCl).
Advertisements
Solution
NaCl
APPEARS IN
RELATED QUESTIONS
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
Give electron dot diagram of the following:
methane
Complete the statement by filling the gaps using appropriate term from the terms given in the bracket.
(slow, coloured, arrow, fast, smell, milky, physical, product, chemical, reactant, covalent, ionic, octet, duplet, exchange, sharing, equality sign)
Sodium chloride is ______ compound while hydrogen chloride is ______ compound.
Among the following compounds identify the compound that has all three bonds (ionic, covalent and coordinate bond).
What do you understand by redox reactions? Explain oxidation and reduction in terms of loss or gain of electrons.
Explain fractional atomic mass. What is the fractional mass of chlorine?
Give reason
Argon does not react.
State the type of bonding in the following molecules.
Water,
State the type of bonding in the following molecules.
Hydrogen chloride
Copy and complete the following table relating to the atomic structure of some elements:
| Element | Atomic Number | Mass Number | Number of protons | Number of Neutrons | Number of Electrons |
| Beryllium | 4 | 9 | |||
| Fluorine | 9 | 10 | |||
| Sodium | 12 | 11 | |||
| Aluminium | 27 | 13 | |||
| Phosphorus | 31 | 15 |
