Advertisements
Advertisements
प्रश्न
Five atoms are labelled V to Z
| Atoms | Mass Number | Atomic Number |
| V | 40 | 20 |
| W | 19 | 9 |
| X | 7 | 3 |
| Y | 16 | 8 |
| Z | 14 | 7 |
- Which one of these atoms
(1) contains 7 protons;
(2) has an electronic configuration 2, 7? - Write down the formula of the compound formed by atoms X and Y.
Advertisements
उत्तर
(i) As Z has atomic number of 7, hence it has 7 protons. Electronic configuration of 2, 7 means the atom has 2 + 7 = 9 electrons. Therefore, element W has electronic configuration 2, 7 since its atomic number is 9.
(ii) Number of electron in X is 3. Therefore, electronic configuration = (2, 1). As X will try to lose 1 electron to attain stable state, hence, X has valency [1+]
Number of electron in Y is 8. Therefore, electronic configuration = (2, 6). As Y will try to gain 2 electrons to attain stable state, hence, Y has valency [2−]
Since the valency of X is 1+ and valency of Y is 2−
Formula of the compound:
X1+ Y2−
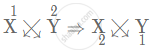
So, we get the formula as X2Y.
APPEARS IN
संबंधित प्रश्न
Separate the following compounds into three categories - ionic, polar and covalent compounds; N2, NH4Cl, NH3, NO, NH4NO3, NCl3.
Name the following:
An element having valency 'zero'
Name the following:
Metal with valency 2
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidation.
State the type of bonding in the following molecules.
Water,
State the type of bonding in the following molecules.
Hydrogen chloride
Explain the following:
Carbon-12 and carbon-14 both show similar chemical properties.
Choose the odd one out and write the reason:
How many atoms of each kind are present in the following molecule:
Chlorine
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is chemically inert.
