Advertisements
Advertisements
प्रश्न
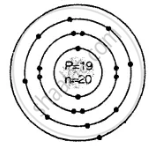
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^39_19K}\]
Advertisements
उत्तर
Geometric atomic structure of:
\[\ce{^39_19K}\]
Number of protons P = 19 = e
A = P + n
39 = 19 + n
∴ n = 39 − 19 = 20
e = 19 (2, 8, 8, 1)
Electronic configuration
2, 8, 8, 1 electronic arrangement
APPEARS IN
संबंधित प्रश्न
Complete the following:
| Sodium | Phosphorus | Carbon | |
| Formula of chloride | |||
| Nature of bonding | |||
| Physical state of chloride |
Explain the following briefly:
Helium does not form He2 molecule
Match the atomic numbers 4, 14, 8, 15 and 19 with each of the following:
- A solid non-metal of valency 3.
- A gas of valency 2.
- A metal of valency 1.
- A non-metal of valency 4.
Why do `""_17^35"CI"` and `""_17^37"CI"` have the same chemical properties? In what respect do these atoms differ?
Name the following:
The shell closest to the nucleus of an atom
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidation.
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of reduction.
In the formation of magnesium chloride (by direct combination between magnesium and chlorine), name the substance that is oxidized and the substance that is reduced.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
