Advertisements
Advertisements
प्रश्न
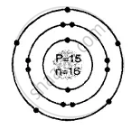
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^31_15P}\]
Advertisements
उत्तर
Geometric atomic structure of:
\[\ce{^31_15P}\]
Number of protons P = 15 = e number of electrons
A = P + n
31 = 15 + n
∴ n = 31 − 15 = 16 number of neutrons
Electronic configuration 2, 8, 5
In K shell = 2 electrons
In L shell = 8
In M shell = 5
APPEARS IN
संबंधित प्रश्न
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms an anion?
State the following is a reduction reaction or oxidation reaction.
\[\ce{A^3+ + e- -> A^2+}\]
Draw orbit structure diagram of sodium chloride (NaCl).
State the type of bonding in the following molecules.
Calcium oxide
Explain the following:
Carbon-12 and carbon-14 both show similar chemical properties.
Identify the following reaction as either oxidation or reduction:
Zn → Zn2+ + 2e-
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^39_19K}\]
Draw the electron distribution diagram for the formation of Carbon di oxide (CO2 ) molecule.
How many electrons are required or released by each atom in water molecule to attain the nearest noble gas configuration?
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
