Advertisements
Advertisements
Question
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^31_15P}\]
Advertisements
Solution
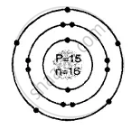
Geometric atomic structure of:
\[\ce{^31_15P}\]
Number of protons P = 15 = e number of electrons
A = P + n
31 = 15 + n
∴ n = 31 − 15 = 16 number of neutrons
Electronic configuration 2, 8, 5
In K shell = 2 electrons
In L shell = 8
In M shell = 5
APPEARS IN
RELATED QUESTIONS
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms an anion?
Complete the statement by filling the gaps using appropriate term from the terms given in the bracket.
(slow, coloured, arrow, fast, smell, milky, physical, product, chemical, reactant, covalent, ionic, octet, duplet, exchange, sharing, equality sign)
Sodium chloride is ______ compound while hydrogen chloride is ______ compound.
In term of electron transfer, define Oxidation
Element X is a metal with a valency 2. Element Y is a non metal with a valency 3.
(a) Write equations to show how X and Y form ions.
(b) If Y is a diatomic gas, write the equation for the direct combination of X and Y to form a compound.
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
How many electrons are required or released by each atom in calcium oxide to attain the nearest noble gas configuration?
Element M forms a chloride with the formula MCl2 which is solid with a high melting point. What type of bond is in MCl2. Write the formula of the compound when M combines with sulphur, oxygen, and nitrogen.
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Draw the electron distribution diagram for the formation of Carbon di oxide (CO2 ) molecule.
An atom X with atomic number 20 combines with atom Y with atomic number 8. Draw the dot structure for the formation of the molecule XY.
