Advertisements
Advertisements
प्रश्न
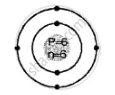
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
Advertisements
उत्तर
Geometric atomic structure of:

K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
संबंधित प्रश्न
State the following is a reduction reaction or oxidation reaction.
\[\ce{Pb -> Pb^2+ + 2e-}\]
Name the following:
An element having valency 'zero'
Choose the correct option
Which of the following is the correct electronic configuration of potassium?
Give the orbital diagram of the following:
Magnesium chloride
State the type of bonding in the following molecules.
Calcium oxide
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
Match the following:
| 1. | Atomic bond | a. | Oxygen and hydrogen |
| 2. | Atoms with different electronegativities | b. | acceptor bond |
| 3. | An atom that accepts electron pair | c. | covalent bond |
| 4. | Rusting of iron | d. | donor atom |
| 5. | An atom that provides electron pair | e. | oxidation |
| f. | ionic bond |
Choose the odd one out and write the reason:
How many electrons are required or released by each atom in chlorine to attain the nearest noble gas configuration?
Elements A, B and C have atomic number 17, 19 and 10 respectively.
Write down the formula of the compound formed by two of the above elements.
