Advertisements
Advertisements
Question
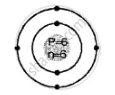
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
Advertisements
Solution
Geometric atomic structure of:

K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
RELATED QUESTIONS
How do atoms attain noble gas configuration?
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
In term of electron transfer, define Oxidation
What type of bond is formed between two atoms, when the electronegative difference between them is zero?
Fill in the blank.
___________ single covalent bonds are formed between the carbon and chlorine atoms.
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
Name the following:
Two atoms having the same number of protons and electrons but different number of neutrons.
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
Explain the following:
Carbon-12 and carbon-14 both show similar chemical properties.
Sodium having atomic number 11, is ready to ______ electron/ electrons to attain the nearest noble gas electronic configuration.
