Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
Advertisements
उत्तर
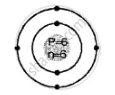
Geometric atomic structure of:

K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
संबंधित प्रश्न
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
State the following is a reduction reaction or oxidation reaction.
\[\ce{Cu -> Cu^2+}\]
Explain fractional atomic mass. What is the fractional mass of chlorine?
Compare :
Sodium atom and sodium ion
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of reduction.
Elements with stable electronic configurations have eight electrons in their valence shell. They are ______.
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Write the basic concept of Kossel – Lewis theory.
