Advertisements
Advertisements
Question
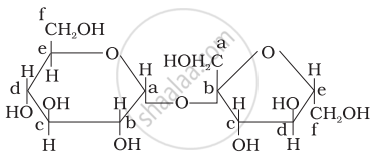
Structure of a disaccharide formed by glucose and fructose is given below. Identify anomeric carbon atoms in monosaccharide units.
Options
‘a’ carbon of glucose and ‘a’ carbon of fructose.
‘a’ carbon of glucose and ‘e’ carbon of fructose.
‘a’ carbon of glucose and ‘b’ carbon of fructose.
‘f’ carbon of glucose and ‘f ’ carbon of fructose.
Advertisements
Solution
‘a’ carbon of glucose and ‘b’ carbon of fructose.
Explanation:
Carbon adjacent to oxygen atom in the cyclic structure of glucose or fructose is known as anomeric carbon. As shown in the structure above ‘a’ and ‘b’ are present at adjacent to oxygen atom. Both carbons differ in configurations of the hydroxyl group.
APPEARS IN
RELATED QUESTIONS
What are the expected products of hydrolysis of lactose?
What are reducing sugars?
What are the hydrolysis products of sucrose?
The number of primary and secondary hydroxyl groups in ribose are -
(a) 1, 3
(b) 2, 3
(c) 3, 1
(d) 3, 2
Glucose gives silver mirror test with Tollen’s reagent. It shows the presence of ____________.
The two functional groups present in a typical carbohydrate are:
Which of the following monosaccharide is pentose?
A carbohydrate that cannot be hydrolysed into simpler units is called ____________.
Reducing sugars reduce ____________.
Which of the following reagent cannot distinguish between glucose and fructose?
Which of the following monosaccharides are present as five membered cyclic structure (furanose structure)?
(i) Ribose
(ii) Glucose
(iii) Fructose
(iv) Galactose
Monosaccharides contain carbonyl group hence are classified, as aldose or ketose. The number of carbon atoms present in the monosaccharide molecule are also considered for classification. In which class of monosaccharide will you place fructose?
What are glycosidic linkages? In which type of biomolecules are they present?
Which monosaccharide units are present in starch, cellulose and glucose and which linkages link these units?
Assertion: Deoxyribose, \[\ce{C5H10O4}\] is not a carbohydrate.
Reason: Carbohydrates are hydrates of carbon so compounds which follow \[\ce{C_x(H2O)_y}\] formula are carbohydrates.
Account for the following:
Glucose is a reducing sugar
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| Carbohydrates are optically active polyhydroxy aldehydes and ketones. They are also called saccharides. All those carbohydrates which reduce Fehling's solution and Tollen's reagent are referred to as reducing sugars. Glucose, the most important source of energy for mammals, is obtained by the hydrolysis of starch. Vitamins are accessory food factors required in the diet. Proteins are the polymers of α-amino acids and perform various structural and dynamic functions in the organisms. Deficiency of vitamins leads to many diseases. |
Answer the following:
(a) The penta-acetate of glucose does not react with Hydroxylamine. What does it indicate? (1)
(b) Why cannot vitamin C be stored in our body? (1)
(c) Define the following as related to proteins: (2)
- Peptide linkage
- Denaturation
OR
(c) Define the following as related to carbohydrates: (2)
- Anomers
- Glycosidic linkage
