Advertisements
Advertisements
Question
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was −15°C, find the temperature to which it was heated.
Advertisements
Solution
Let the original volume (V) = 1 and
the original pressure (P) = 1 and
the temperature given (T) = −15°C.
= −15 + 273
= 258 K
V1 or new volume after heating = original volume + 50% of original volume
= `1 + 1 xx 50/100`
= `1 + 1/2`
= `3/2`
P1 or decreased pressure = 60%
`1 xx 60/100 = 0.6`
T1 = to be calculated
`(PV)/T = (P_1 V_1)/T_1`
`(1 xx 1)/258 = 3/2 xx 0.6/T_1`
T1 = 232.2
T1 = 232.2 − 273
= −40.8°C
APPEARS IN
RELATED QUESTIONS
Give the assumptions of the kinetic molecular theory.
Explain Charles's law on the basis of the kinetic theory of matter.
State the law which is represented by the following graph:
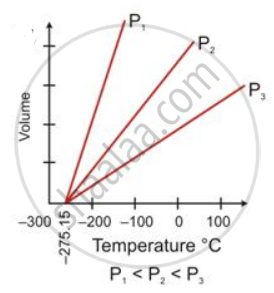
How did Charles's law lead to the concept of an absolute scale of temperature?
State the following:
Ice point in absolute temperature
Choose the correct answer:
The volume-temperature relationship is given by
An LPG cylinder can withstand a pressure of 14.9 atmospheres. The pressure gauge of the cylinder indicates 12 atmospheres at 27°C. Because of a sudden fire in the building, the temperature rises. At what temperature will the cylinder explode?
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
