Advertisements
Advertisements
Question
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Advertisements
Solution
(i) stoichiometric defect
(ii) dislocation defect
Explanation:
In Frenkel defect, dislocation of cations takes place and there is no change in stoichiometry of the crystal.
APPEARS IN
RELATED QUESTIONS
Explain the following term with suitable examples:
Frenkel defect
Explain the following term with a suitable example:
Interstitials
What type of defect is shown by NaCl in
stoichiometric defects, and
Out of NaCl and AgCl, which one shows Frenkel defect and why?
Dislocation defect is also known as ____________.
Type of stoichiometric defect shown by ZnS is ____________.
The following diagram shows:
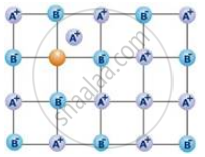
Which is not correct about the Schottky defects?
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Alkali halids do not show Frenkel defect because ____________.
Which of the following crystals does not exhibit Frenkel defect?
Cations are present in the interstitial sites in ______.
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Why is FeO (s) not formed in stoichiometric composition?
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
The incorrect statement about the imperfections in solids is ______.
Which type of ‘defect’ has the presence of cations in the interstitial sites?
