Advertisements
Advertisements
प्रश्न
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Advertisements
उत्तर
(i) stoichiometric defect
(ii) dislocation defect
Explanation:
In Frenkel defect, dislocation of cations takes place and there is no change in stoichiometry of the crystal.
APPEARS IN
संबंधित प्रश्न
Explain the following term with suitable examples:
Frenkel defect
Explain the following term with a suitable example:
Interstitials
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
What type of defect is shown by NaCl in
stoichiometric defects, and
non-stoichiometric defects?
Ionic solids containing large differences in sizes of ions show ____________.
Point defect is also known as ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
The following diagram shows:
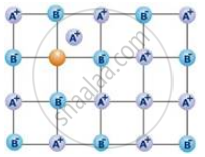
Which is not correct about the Schottky defects?
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Alkali halids do not show Frenkel defect because ____________.
Silver halides generally show:
In a Schottky defect ____________.
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
The incorrect statement about the imperfections in solids is ______.
Which type of ‘defect’ has the presence of cations in the interstitial sites?
