Advertisements
Advertisements
प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Advertisements
उत्तर
Schottky defect is shown by the ionic solids having very small difference in their cationic and anionic radius whereas Frenkel defect is shown by ionic solids having large difference in their cationic and anionic radius. In this case the radius of both Na+ and Cl- have very small difference in their radius as a result of it NaCl will show schottky defect.
APPEARS IN
संबंधित प्रश्न
Explain the following term with a suitable example:
Interstitials
non-stoichiometric defects?
Answer the following question.
What type of stoichiometric defect is shown by ZnS and why?
Defect shown by non-ionic solids is ____________.
Defects in solids can be studied using
Dislocation defect is also known as ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
The following diagram shows:
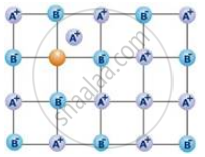
Alkali halids do not show Frenkel defect because ____________.
Which of the following defects is also known as dislocation defect?
