Advertisements
Advertisements
Question
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Options
A and B
C and D
A and C
B and D
Advertisements
Solution
A and B
Explanation:
AgBr shows both Schottky and Frenkel defects.
In AgBr, both Ag+ and Br ions are absent from the lattice causing Schottky defect. However, Ag+ ions are mobile so they have a tendency to move aside the lattice and trapped in interstitial site, hence causing Frenkel defect.
APPEARS IN
RELATED QUESTIONS
Explain the following term with a suitable example:
Interstitials
Examine the given defective crystal:

Answer the following questions :
(i) What type of stoichiometric defect is shown by the crystal?
(ii) How is the density of the crystal affected by this defect?
(iii) What type of ionic substances show such defect?
Out of NaCl and AgCl, which one shows Frenkel defect and why?
Defects in solids can be studied using
Dislocation defect is also known as ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
Type of stoichiometric defect shown by ZnS is ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
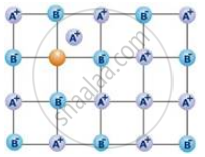
Schottky defect defines imperfection in the lattice structure of ____________.
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Alkali halids do not show Frenkel defect because ____________.
What is the effect of Frenkel defect on the density of ionic solids?
Cations are present in the interstitial sites in ______.
Which of the following defects is also known as dislocation defect?
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
Match the defects given in Column I with the statements in given Column II.
| Column I | Column II |
| (i) Simple vacancy defect | (a) shown by non-ionic solids and increases density of the solid. |
| (ii) Simple interstitial defect | (b) shown by ionic solids and decreases density of the solid. |
| (iii) Frenkel defect | (c) shown by non ionic solids and density of the solid decreases. |
| (iv) Schottky defect | (d) shown by ionic solids and density of the solid remains the same. |
