Advertisements
Advertisements
Question
Which of the following defects is also known as dislocation defect?
Options
Frenkel defect
Schottky defect
Non-stoichiometric defect
Simple interstitial defect
Advertisements
Solution
Frenkel defect
Explanation:
In Frenkel defect, some cations occupy interstitial site and hence it is also called dislocation defect.
APPEARS IN
RELATED QUESTIONS
Explain the following term with suitable examples:
Frenkel defect
Explain the following term with a suitable example:
Interstitials
Defect shown by non-ionic solids is ____________.
Point defect is also known as ____________.
Dislocation defect is also known as ____________.
The following diagram shows:
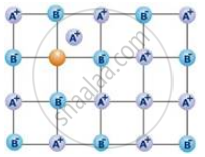
Which is not correct about the Schottky defects?
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Alkali halids do not show Frenkel defect because ____________.
Cations are present in the interstitial sites in ______.
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Why is FeO (s) not formed in stoichiometric composition?
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
Schottky defect is noticed in ______
Given below are two statements:
Statements I: Frenkel defects are vacancy as well as interstitial defects.
Statements II: Frenkel defect leads of colour in ionic solids due to the presence of F-centers.
Choose the most appropriate answer for the statements from the options given below:
The incorrect statement about the imperfections in solids is ______.
