Advertisements
Advertisements
Question
Explain the following term with a suitable example:
Interstitials
Advertisements
Solution
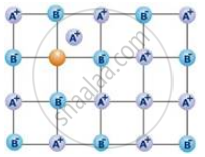
- When certain constituent particles, such as atoms or molecules, occupy interstitial positions, we refer to a crystal as having an interstitial defect.
- As shown in the figure, the presence of interstitial particles increases the substance’s density.

- Non-ionic solids exhibit interstitial flaws and vacancies.
- Ionic solids do not have simple vacancies or interstitial flaws as previously described because they must always maintain electrical neutrality.
- Rather, they display additional flaws referred to as Frenkel and Schottky faults.
- An example of interstitials is Carbon in Iron (Steel); Carbon atoms occupy interstitial sites in the iron lattice, making the metal stronger and harder.
RELATED QUESTIONS
Explain the following term with suitable examples:
Frenkel defect
Defects in solids can be studied using
Dislocation defect is also known as ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
Schottky defect defines imperfection in the lattice structure of ____________.
Which is not correct about the Schottky defects?
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Which of the following crystals does not exhibit Frenkel defect?
What is the effect of Frenkel defect on the density of ionic solids?
Which of the following statements are not true?
(i) Vacancy defect results in a decrease in the density of the substance.
(ii) Interstitial defects results in an increase in the density of the substance.
(iii) Impurity defect has no effect on the density of the substance.
(iv) Frankel defect results in an increase in the density of the substance.
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
If NaCl is doped with 10–4 mole percent of SrCl2 the concentration of cation vacancies will be
Given below are two statements:
Statements I: Frenkel defects are vacancy as well as interstitial defects.
Statements II: Frenkel defect leads of colour in ionic solids due to the presence of F-centers.
Choose the most appropriate answer for the statements from the options given below:
