Advertisements
Advertisements
Question
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
Advertisements
Solution
(ii) Vacancy defect
(iv) Schottky defect
Explanation:
Vacancy and Schottky defect which lead to decrease the density both are the types of a stoichiometric defect. In case of Frenkel defect and interstitial defect, there is no change in density of substance.
APPEARS IN
RELATED QUESTIONS
Explain the following term with suitable examples:
Frenkel defect
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
What type of defect is shown by NaCl in
stoichiometric defects, and
Answer the following question.
What type of stoichiometric defect is shown by ZnS and why?
Defect shown by non-ionic solids is ____________.
Point defect is also known as ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
The following diagram shows:
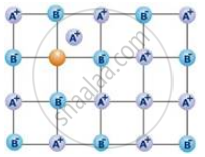
Schottky defect defines imperfection in the lattice structure of ____________.
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
In a Schottky defect ____________.
Which of the following statements are not true?
(i) Vacancy defect results in a decrease in the density of the substance.
(ii) Interstitial defects results in an increase in the density of the substance.
(iii) Impurity defect has no effect on the density of the substance.
(iv) Frankel defect results in an increase in the density of the substance.
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Match the defects given in Column I with the statements in given Column II.
| Column I | Column II |
| (i) Simple vacancy defect | (a) shown by non-ionic solids and increases density of the solid. |
| (ii) Simple interstitial defect | (b) shown by ionic solids and decreases density of the solid. |
| (iii) Frenkel defect | (c) shown by non ionic solids and density of the solid decreases. |
| (iv) Schottky defect | (d) shown by ionic solids and density of the solid remains the same. |
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
