Advertisements
Advertisements
Question
Explain the following term with suitable examples:
Frenkel defect
Advertisements
Solution
This defect arises when an ion (generally a cation) leaves its lattice site and occupies an interstitial site.

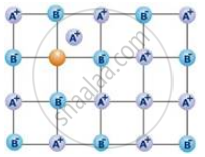
The void or empty space between the lattice points is known as an interstitial site. Fig. depicts a Frenkel flaw. It is evident from the image that instead of having its own proper location in the lattice, one of the A+ ions occupies a spot in the interstitial space. As seen in the illustration, this results in a hole being made in the lattice. Because the number of positive ions equals the number of negative ions, the crystal is nonetheless neutral despite this defect, maintaining the compound’s stoichiometry and electrical neutrality.
Frenkel flaws do not impact the crystal’s density. They conduct electricity in crystals and cause diffusion in solids. The presence of Ag+ ions in the interstitial region of AgBr crystals creates photographic images when exposed to light. Frenkel flaws raise the dielectric constant of crystals. Frenkel flaws cause charges to move closer within the crystal.
Frenkel defects are common in silver halides, e.g., AgCl, AgBr and Agl. They are also found in ZnS crystals.
RELATED QUESTIONS
Explain the following term with a suitable example:
Interstitials
Examine the given defective crystal:

Answer the following questions :
(i) What type of stoichiometric defect is shown by the crystal?
(ii) How is the density of the crystal affected by this defect?
(iii) What type of ionic substances show such defect?
What type of defect is shown by NaCl in
stoichiometric defects, and
Ionic solids containing large differences in sizes of ions show ____________.
Point defect is also known as ____________.
Frenkel defects are not found in alkali metal halides because ____________.
Type of stoichiometric defect shown by ZnS is ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Silver halides generally show:
What is the effect of Frenkel defect on the density of ionic solids?
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
Why is FeO (s) not formed in stoichiometric composition?
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion: In any ionic solid (MX) with Schottky defects, the number of positive and negative ions are same.
Reason: Equal number of cation and anion vacancies are present.
Schottky defect is noticed in ______
Which type of ‘defect’ has the presence of cations in the interstitial sites?
