Advertisements
Advertisements
Question
For a reaction : 
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Advertisements
Solution
(i) This reaction is catalysed by Pt at high pressure. So, it is a zero-order reaction with molecularity 2.
(ii) The rate law expression for this reaction is
Rate = k
Hence, the unit of k is mol L−1 s−1.
APPEARS IN
RELATED QUESTIONS
Define “zero order reaction”.
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:

(i) Predict the order of the reaction.
(ii) What is the slope of the curve?
(iii) Write the unit of rate constant for this reaction.
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
Define the following term:
Pseudo first-order reaction
For which type of reactions, order and molecularity have the same value?
In a reaction if the concentration of reactant A is tripled, the rate of reaction becomes twenty seven times. What is the order of the reaction?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?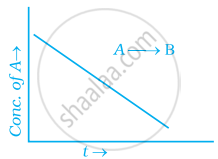
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
For a reaction \[\ce{Cl2l(g) + 2No(g) -> 2NaCl(g)}\] the rate law is expressed as rate= K[Cl2] [No]2 what is the order of the reaction?
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
