Advertisements
Advertisements
Question
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
Advertisements
Solution
Gattermann-Koch reaction is formylation (introduction of-CHO group) to the benzene nucleus. Friedel-Crafts acylation reaction is introduction of RCO-group in the benzene ring.

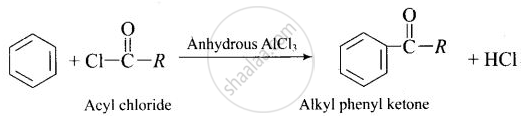
In Friedel-Crafts acylation reactions, the benzene is treated with an acid chloride in presence of anhydrous \[\ce{AlCl3}\]. So, Gattermann-Koch reaction can be considered similar to Friedel-Crafts acylation reaction.
APPEARS IN
RELATED QUESTIONS
Write the chemical equations to illustrate the following name reactions : Rosenmund reduction
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde.
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzaldehyde
When 0.4 g of acetic acid is dissolved in 40 g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol−1, at. wt. C = 12, H = 1, O = 16)
Aldehydes are produced on reduction of the following by DIBAL-H:
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
Ethylbenzene is generally prepared by acetylation of benzene followed by reduction and not by direct alkylation. Think of a possible reason.
Match the acids given in Column I with their correct IUPAC names given in Column II.
| Column I (Acids) |
Column II (IUPAC names) |
||
| (i) | Phthalic acid | (a) | Hexane-1,6-dioic acid |
| (ii) | Oxalic acid | (b) | Benzene-1,2-dicarboxylic acid |
| (iii) | Succinic acid | (c) | Pentane-1,5-dioic acid |
| (iv) | Adipic acid | (d) | Butane-1,4-dioic acid |
| (v) | Glutaric acid | (e) | Ethane-1,2-dioic acid |
An alkene ‘A’ (Mol. formula \[\ce{C5H10}\]) on ozonolysis gives a mixture of two compounds ‘B’ and ‘C’. Compound ‘B’ gives positive Fehling’s test and also forms iodoform on treatment with \[\ce{I2}\] and \[\ce{NaOH}\]. Compound ‘C’ does not give Fehling’s test but forms iodoform. Identify the compounds A, B and C. Write the reaction for ozonolysis and formation of iodoform from B and C.
In the chromyl chloride test, the final step results in the formation of a yellow precipitate of the following:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Cl}
\end{array}\] obtained by chlorination of n-butane, will be
When 2 – hydroxyl benzoic acid distilled with zinc dust, it give
The intermediate compound ‘X’ in the following chemical reaction is:

Convert the following:
Benzoic acid to Benzaldehyde.
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
Reagent used to convert allyl alcohol to acrolein is ______.
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
