Advertisements
Advertisements
Question
Match the acids given in Column I with their correct IUPAC names given in Column II.
| Column I (Acids) |
Column II (IUPAC names) |
||
| (i) | Phthalic acid | (a) | Hexane-1,6-dioic acid |
| (ii) | Oxalic acid | (b) | Benzene-1,2-dicarboxylic acid |
| (iii) | Succinic acid | (c) | Pentane-1,5-dioic acid |
| (iv) | Adipic acid | (d) | Butane-1,4-dioic acid |
| (v) | Glutaric acid | (e) | Ethane-1,2-dioic acid |
Match the Columns
Advertisements
Solution
| Column I (Acids) |
Column II (IUPAC names) |
||
| (i) | Phthalic acid | (b) | Benzene-1,2-dicarboxylic acid |
| (ii) | Oxalic acid | (e) | Ethane-1,2-dioic acid |
| (iii) | Succinic acid | (d) | Butane-1,4-dioic acid |
| (iv) | Adipic acid | (a) | Hexane-1,6-dioic acid |
| (v) | Glutaric acid | (c) | Pentane-1,5-dioic acid |
Explanation:
| Acids | IUPAC names | Structure |
| (i) Phthalic acid | Benzene-1,2-dicarboxylic acid | 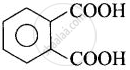 |
| (ii) Oxalic acid | Ethane-1,2-dioic acid | \[\begin{array}{cc} \ce{COOH}\\ |\phantom{......}\\ \ce{COOH} \end{array}\] |
| (iii) Succinic acid | Butane-1,4-dioic acid |  |
| (iv) Adipic acid | Hexane-1,6-dioic acid | 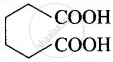 |
| (v) Glutaric acid | Pentane-1,5-dioic acid |  |
shaalaa.com
Is there an error in this question or solution?
