Advertisements
Advertisements
Question
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Cl}
\end{array}\] obtained by chlorination of n-butane, will be
Options
`l` - form
d - form
Meso form
Racemic mixture
MCQ
Advertisements
Solution
Racemic mixture
Explanation:
Chlorination ofn-butane takes place via free radical formation i.e., \[\ce{Cl2 ->[hv] C\overset{\bullet}{l} + C\overset{\bullet}{l}}\]
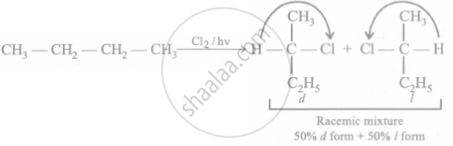
\[\ce{C\overset{\bullet}{l}}\] may attack on either side and give a racemic mixture of 2-chlorbutane which contain 50% d - form and 50% `l` - form.
shaalaa.com
Is there an error in this question or solution?
