Advertisements
Advertisements
प्रश्न
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
टिप्पणी लिखिए
Advertisements
उत्तर
Gattermann-Koch reaction is formylation (introduction of-CHO group) to the benzene nucleus. Friedel-Crafts acylation reaction is introduction of RCO-group in the benzene ring.

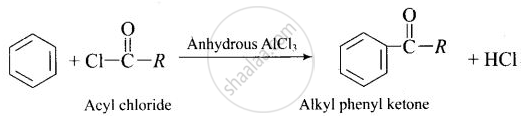
In Friedel-Crafts acylation reactions, the benzene is treated with an acid chloride in presence of anhydrous \[\ce{AlCl3}\]. So, Gattermann-Koch reaction can be considered similar to Friedel-Crafts acylation reaction.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
