Advertisements
Advertisements
Question
Answer in brief.
Give a simple chemical test to distinguish between ethanol and ethyl bromide.
Advertisements
Solution
i. Ethanol on reaction with a very strong base like alkali metal Na or K gives sodium or potassium ethoxide with the liberation of hydrogen gas.
\[\ce{2CH3CH2 - OH + 2Na -> 2CH3CH2O^-Na+ + H2_{(g)}↑}\]
ii. Ethyl bromide on reaction with sodium metal does not liberate hydrogen gas.
\[\ce{2CH3CH2Br + 2Na -> CH3CH2 - CH2CH3 + 2NaBr}\]
APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Write two points of difference between the properties of phenol and ethyl alcohol.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Write IUPAC name of crotonyl alcohol.
Write chemical equation of acetyl chloride with phenol
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Which of the following compounds is obtained, when phenol react with bromine water?
Phenol is obtained from cumene ____________.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
Identify 'A' and 'B' in the following series of reactions.
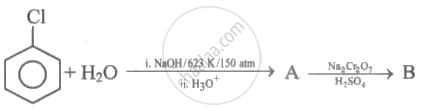
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
+I effect of alkyl groups in alcohols increases the stability of ____________.
Phenoxide ion is more stable than phenol due to the ____________.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
Which of the following conversion explains the acidic nature of alcohols?
Which of the following on oxidation yields ethyl methyl ketone?
Identify the alcohol that reacts immediately with Lucas reagent?
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
Which among the following is not the method of preparation of phenol?
The product C in the following reaction is

The chemical test that distinguish between benzoic acid and phenol is ______.
Arrange the following compounds in an increasing order of their solubility in water:
Propene can be converted into propan-1-ol by:
What is the commercial method of preparation of phenol?
The C−O−C angle in ether is about ______.
Which of the following is primary benzylic alcohol?
