Advertisements
Advertisements
Question
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Advertisements
Solution
- Dilute HNO3: Phenol reacts with dilute nitric acid at low temperature to give a mixture of ortho- and para-nitrophenol.

- Conc. H2SO4: At room temperature (298 K), phenol reacts with concentrated sulphuric acid to form o-phenolsulphonic acid.

- Br2 in CS2: When a reaction is carried out in a solvent of lower polarity like CS2 a mixture of ortho- and para-bromophenol is formed.

RELATED QUESTIONS
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Write two points of difference between the properties of phenol and ethyl alcohol.
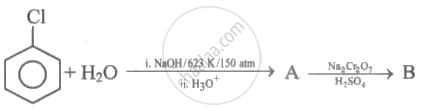
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Reaction between hot conc. HI and anisole gives ______________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Which alcohol is difficult to oxidise?
____________ is used for silvering mirrors.
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Which of following elements does not react with hot concentrated sulphuric acid?
Identify 'A' and 'B' in the following series of reactions.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Phenoxide ion is more stable than phenol due to the ____________.
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Which of the following conversion explains the acidic nature of alcohols?
The number of σ bonds in carbolic acid is ______.
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Identify the role of PDC from the following.
Ethyl alcohol can be prepared from a Grignard reagent by the reaction of ______.
Name the catalyst used in commercial method of preparation of phenol.
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
The chemical test that distinguish between benzoic acid and phenol is ______.
Convert the following :
cumene to phenol.
The C−O−C angle in ether is about ______.
Which of the following is trihydric phenol?
