Advertisements
Advertisements
Question
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Advertisements
Solution
Decreasing order of acid strength:
CH3OH > CH3–CH2–OH > CH3–CH(OH)–CH3 > (CH3)3–C–OH
APPEARS IN
RELATED QUESTIONS
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
Write IUPAC name of crotonyl alcohol.
How will you bring about the following conversions?
isopropyl alcohol to acetone
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
____________ is used for silvering mirrors.
Phenol is obtained from cumene ____________.
Bromination of phenol, will NOT give:
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which among the following compounds is used to prepare solid fuel using calcium acetate?
The number of σ bonds in carbolic acid is ______.
Identify the alcohol that reacts immediately with Lucas reagent?
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Convert the following :
cumene to phenol.
The structure of A in the given reaction is:
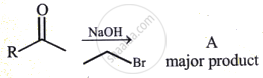
What is the name of isopropyl alcohol according to the carbinol system?
Which of the following is primary benzylic alcohol?
