Advertisements
Advertisements
प्रश्न
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Advertisements
उत्तर
Decreasing order of acid strength:
CH3OH > CH3–CH2–OH > CH3–CH(OH)–CH3 > (CH3)3–C–OH
APPEARS IN
संबंधित प्रश्न
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
Write the reaction to get aspirin from salicylic acid.
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Write Kolbe’s reaction.
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Phenol is obtained from cumene ____________.
Sodium metal with ethyl alcohol gives __________ gas.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which following reagent is used to obtain alkene from alcohol?
Which of the following compounds does NOT react with sodium metal?
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
The acid, which contains both -OH and -COOR groups is ______.
Which of the following compounds has lowest boiling point?
Which among the following is not the method of preparation of phenol?
Arrange the following compounds in an increasing order of their solubility in water:
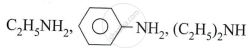
Explain: Phenols are acid while alcohol is neutral.
Which of the following is trihydric phenol?
