Advertisements
Advertisements
प्रश्न
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
विकल्प
acetaldehyde
acetic acid
ethane
acetone
Advertisements
उत्तर
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of acetic acid
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Reaction between hot conc. HI and anisole gives ______________
Write the reaction to get aspirin from salicylic acid.
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Which of the following compounds does not react with bromine in alkaline medium?
Bromination of phenol, will NOT give:
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
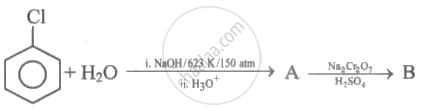
Identify 'A' and 'B' in the following series of reactions.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
In Dow process, chlorobenzene is converted into phenol by ____________.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
Which isomer of C4H10O is optically active?
Which among the following is allylic secondary alcohol?
Which of the following compounds does NOT react with sodium metal?
Which of the following on oxidation yields ethyl methyl ketone?
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
The acid, which contains both -OH and -COOR groups is ______.
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
The product C in the following reaction is

The major product obtained in the following reaction is

Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
Convert the following :
cumene to phenol.
What is the name of isopropyl alcohol according to the carbinol system?
Which of the following is trihydric phenol?
