Advertisements
Advertisements
प्रश्न
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Advertisements
उत्तर
- Dilute HNO3: Phenol reacts with dilute nitric acid at low temperature to give a mixture of ortho- and para-nitrophenol.

- Conc. H2SO4: At room temperature (298 K), phenol reacts with concentrated sulphuric acid to form o-phenolsulphonic acid.

- Br2 in CS2: When a reaction is carried out in a solvent of lower polarity like CS2 a mixture of ortho- and para-bromophenol is formed.

संबंधित प्रश्न
Write two points of difference between the properties of phenol and ethyl alcohol.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Write the name of major product when anisole reacts HI at 398 K
Write Kolbe’s reaction.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Write chemical equation of acetyl chloride with phenol
____________ is used for silvering mirrors.
In phenols, −OH group is attached to ___________ hybridised carbon.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
In Dow process, chlorobenzene is converted into phenol by ____________.
____________ will NOT undergo acetylation.
Phenoxide ion is more stable than phenol due to the ____________.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
(I)

(II) \[\ce{P + Zn ->[\Delta] Q}\]
(III) \[\ce{P ->[Na2Cr2O7][H2SO4] R}\]
Q and R are respectively:
Which of the following conversion explains the acidic nature of alcohols?
Which isomer of C4H10O is optically active?
Which of the following compounds does NOT react with sodium metal?
Which of the following on oxidation yields ethyl methyl ketone?
Identify the role of PDC from the following.
The acid, which contains both -OH and -COOR groups is ______.
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
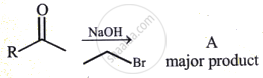
The major product obtained in the following reaction is

What is the action of nitrous acid on aniline?
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Propene can be converted into propan-1-ol by:
The structure of A in the given reaction is: