Advertisements
Advertisements
प्रश्न
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
Advertisements
उत्तर
2-Methylpropan-2-ol undergoes dehydration when passed over hot copper at 573 K.
\[\begin{array}{cc}
\ce{H3C - C(OH) - CH3 ->[Cu/573K][dehydration] H3C - C = CH2}\\
\phantom{}|\phantom{...............................}|\phantom{.}\\
\phantom{....}\ce{\underset{\text{2-Methylpropan-2-ol}}{CH3}\phantom{..................}\underset{\text{2-Methylpropene}}{CH3}}\phantom{....}
\end{array}\]
संबंधित प्रश्न
Answer in one sentence/ word.
Write the IUPAC name of alcohol having molecular formula C4H10O which is resistant towards oxidation.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Write the reactions for the preparation of carbolic acid from aniline.
Which alcohol is difficult to oxidise?
In phenols, −OH group is attached to ___________ hybridised carbon.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
Which of the following is INCORRECT regarding phenol?
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which following reagent is used to obtain alkene from alcohol?
Which of the following alcohols is least soluble in water?
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Identify the product obtained when phenol is treated with bromine water?
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
Which of the following compounds has lowest boiling point?
What is the action of nitrous acid on aniline?
Arrange the following compounds in an increasing order of their solubility in water:
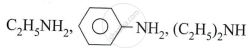
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
Convert the following :
cumene to phenol.
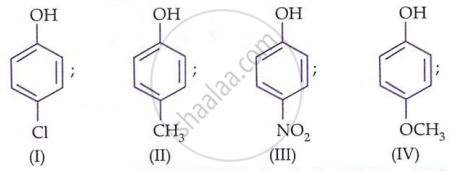
Arrange the following compounds in order of decreasing acidity:
What is the name of isopropyl alcohol according to the carbinol system?
