Advertisements
Advertisements
प्रश्न
How will you bring about the following conversions?
isopropyl alcohol to acetone
Advertisements
उत्तर
When isopropyl alcohol is heated at 573 K in the presence of Cu, acetone is formed.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[Cu/573K][Oxidation] CH3 - C - CH3}\\
\phantom{...}|\phantom{..........................}||\phantom{...}\\
\ce{\underset{\text{Isopropyl alcohol}}{OH}\phantom{..................}\underset{\text{Acetone}}{O}\phantom{...}}\\
\end{array}\]
संबंधित प्रश्न
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Write two points of difference between the properties of phenol and ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Write the reaction to get aspirin from salicylic acid.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Which alcohol is difficult to oxidise?
Sodium metal with ethyl alcohol gives __________ gas.
Which of following elements does not react with hot concentrated sulphuric acid?
The number of isomeric alcohols possible with the formula C4H10O is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
+I effect of alkyl groups in alcohols increases the stability of ____________.
Which among the following compounds is used to prepare solid fuel using calcium acetate?
The number of σ bonds in carbolic acid is ______.

Product (B) in this reaction is:
The product C in the following reaction is

Arrange the following compounds in an increasing order of their solubility in water:
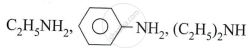
What is the commercial method of preparation of phenol?
The C−O−C angle in ether is about ______.
What is the name of isopropyl alcohol according to the carbinol system?
