Advertisements
Advertisements
प्रश्न
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Advertisements
उत्तर
The difference in the acidic character of phenols and alcohol is due to the difference in reactivity of these compounds towards the ionization of the O–H bond. This can be explained as follows:
i. Ionization of alcohols is represented by the following equilibrium
\[\ce{\underset{\text{Alcohol}}{R - OH} + H2O ⇌ \underset{\text{Alkoxide}}{R - O-} + H3O+}\]
The electron-donating inductive effect (+I effect) of the alkyl group destabilizes the alkoxide ion (the conjugate base of alcohol). As a result, alcohol does not ionize much in the water, and behaves like a neutral compound in an aqueous medium.
ii. Ionization of phenol is represented by the following equilibrium


Phenoxide ion, the conjugate base of phenol, is resonance stabilized by delocalization of the negative charge. Therefore, phenol ionizes in an aqueous medium to a moderate extent. Thus phenols are more acidic than ethyl alcohol.
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Write the name of major product when anisole reacts HI at 398 K
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
Write Kolbe’s reaction.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
Cumene is used in the commercial method for the manufacture of ____________.
Identify 'A' and 'B' in the following series of reactions.
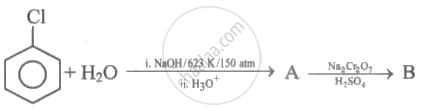
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
In Dow process, chlorobenzene is converted into phenol by ____________.
Which of the following is INCORRECT regarding phenol?
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.

Identify reagents X, Y and Z.
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which of the following alcohols is least soluble in water?
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Identify the alcohol that reacts immediately with Lucas reagent?
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.

Product (B) in this reaction is:
Which of the following compounds has lowest boiling point?
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
The product C in the following reaction is

The order of reactivity of hydrogen halides with ether is as follows.
The major product obtained in the following reaction is

Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
Propene can be converted into propan-1-ol by:
