Advertisements
Advertisements
प्रश्न
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Advertisements
उत्तर
Carbon dioxide
संबंधित प्रश्न
Answer in one sentence/ word.
Write the IUPAC name of alcohol having molecular formula C4H10O which is resistant towards oxidation.
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
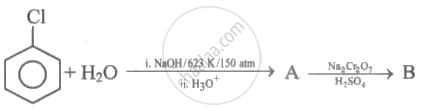
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Write the reaction to get aspirin from salicylic acid.
Write the reaction involved in the preparation of phenol from benzene sulphonic acid.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Write the reactions for the preparation of carbolic acid from aniline.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
Which of the following compounds does not react with bromine in alkaline medium?
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
Identify 'A' and 'B' in the following series of reactions.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
____________ will NOT undergo acetylation.

Identify reagents X, Y and Z.
Which of the following reagents is used to convert phenol to benzene?
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Which among the following is allylic secondary alcohol?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.

Product (B) in this reaction is:
Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
