Advertisements
Advertisements
प्रश्न
Answer in brief.
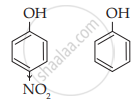
Explain why p-nitrophenol is a stronger acid than phenol.
Advertisements
उत्तर
- In ρ-nitrophenol, nitro group (NO2) is an electron-withdrawing group present at para position which enhances the acidic strength (- I effect). The O – H bond is under strain and release of proton (H+) becomes easy. Further, ρ-nitrophenol ion is more stabilised due to resonance.
- Since the absence of electron-withdrawing group (like –NO2) in phenol at ortho and para position, the acidic strength of phenol is less than that of ρ-nitrophenol.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
Which alcohol is difficult to oxidise?
____________ is used for silvering mirrors.
Which of the following compounds does not react with bromine in alkaline medium?
Phenol is obtained from cumene ____________.
Phenol is ____________.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
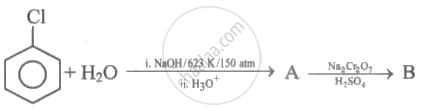
Identify 'A' and 'B' in the following series of reactions.
____________ will NOT undergo acetylation.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.

Identify reagents X, Y and Z.
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Identify the role of PDC from the following.
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
Which among the following is not the method of preparation of phenol?
The major product obtained in the following reaction is

What is the action of nitrous acid on aniline?
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
What is the name of isopropyl alcohol according to the carbinol system?
