Advertisements
Advertisements
Question
Answer in brief.
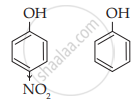
Explain why p-nitrophenol is a stronger acid than phenol.
Advertisements
Solution
- In ρ-nitrophenol, nitro group (NO2) is an electron-withdrawing group present at para position which enhances the acidic strength (- I effect). The O – H bond is under strain and release of proton (H+) becomes easy. Further, ρ-nitrophenol ion is more stabilised due to resonance.
- Since the absence of electron-withdrawing group (like –NO2) in phenol at ortho and para position, the acidic strength of phenol is less than that of ρ-nitrophenol.
APPEARS IN
RELATED QUESTIONS
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Reaction between hot conc. HI and anisole gives ______________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
How will you bring about the following conversions?
isopropyl alcohol to acetone
Phenol is obtained from cumene ____________.
Sodium metal with ethyl alcohol gives __________ gas.
Phenol is ____________.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
Cumene is used in the commercial method for the manufacture of ____________.
Phenoxide ion is more stable than phenol due to the ____________.
The most resistant alcohol towards oxidation reaction is:

Identify reagents X, Y and Z.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which isomer of C4H10O is optically active?
Which of the following alcohols is least soluble in water?
Identify the alcohol that reacts immediately with Lucas reagent?
Identify the compound having highest boiling point from the following?
Identify the role of PDC from the following.
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.

Product (B) in this reaction is:
What is the product obtained in Reimer - Tiemann reaction?
What is the action of nitrous acid on aniline?
Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Which of the following is primary benzylic alcohol?
