Advertisements
Advertisements
Question
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Advertisements
Solution
- Concentrated nitric acid in the presence of conc. H2SO4 is used to convert phenol into picric acid.
- Chromic anhydride or sodium dichromate in the presence of H2SO4 is used to convert phenol into p-benzoquinone.
APPEARS IN
RELATED QUESTIONS
Answer in one sentence/ word.
Write the IUPAC name of alcohol having molecular formula C4H10O which is resistant towards oxidation.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Reaction between hot conc. HI and anisole gives ______________
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
Write the reactions for the preparation of carbolic acid from aniline.
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
Identify 'A' and 'B' in the following series of reactions.
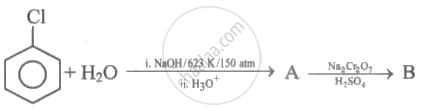
In Dow process, chlorobenzene is converted into phenol by ____________.
+I effect of alkyl groups in alcohols increases the stability of ____________.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which of the following conversion explains the acidic nature of alcohols?
Which isomer of C4H10O is optically active?
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following on oxidation yields ethyl methyl ketone?
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Ethyl alcohol can be prepared from a Grignard reagent by the reaction of ______.
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
The product C in the following reaction is

The chemical test that distinguish between benzoic acid and phenol is ______.
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
