Advertisements
Advertisements
Question
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Advertisements
Solution
Isopropylbenzene: This is the commercial method of preparation of phenol. Isopropylbenzene (Cumene) on air oxidation in presence of Conaphthenate gives cumene hydroperoxide, which on decomposition with dilute acid gives phenol with acetone as a valuable by-product.

APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Write IUPAC name of crotonyl alcohol.
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Write the reactions for the preparation of carbolic acid from aniline.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
In phenols, −OH group is attached to ___________ hybridised carbon.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
Which of following elements does not react with hot concentrated sulphuric acid?
The number of isomeric alcohols possible with the formula C4H10O is ____________.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
____________ will NOT undergo acetylation.
Phenoxide ion is more stable than phenol due to the ____________.
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify product B in the following conversion?
\[\ce{Phenol ->[NaOH] A ->[CH3I] B}\]
The major product obtained in the following reaction is

What is the action of nitrous acid on aniline?
Arrange the following compounds in an increasing order of their solubility in water:
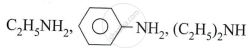
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
Explain: Phenols are acid while alcohol is neutral.
