Advertisements
Advertisements
प्रश्न
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Advertisements
उत्तर
Isopropylbenzene: This is the commercial method of preparation of phenol. Isopropylbenzene (Cumene) on air oxidation in presence of Conaphthenate gives cumene hydroperoxide, which on decomposition with dilute acid gives phenol with acetone as a valuable by-product.

APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Answer in brief.
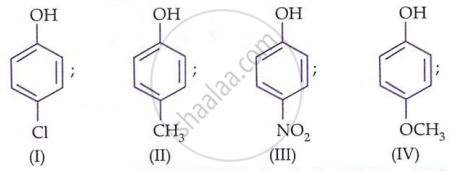
Explain why p-nitrophenol is a stronger acid than phenol.
Answer in brief.
Give a simple chemical test to distinguish between ethanol and ethyl bromide.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Reaction between hot conc. HI and anisole gives ______________
Write Kolbe’s reaction.
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
What is the action of conc. H2SO4 on carbolic acid at 373 K.
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
____________ is used for silvering mirrors.
In phenols, −OH group is attached to ___________ hybridised carbon.
Sodium metal with ethyl alcohol gives __________ gas.
Cumene is used in the commercial method for the manufacture of ____________.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
+I effect of alkyl groups in alcohols increases the stability of ____________.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
(I)

(II) \[\ce{P + Zn ->[\Delta] Q}\]
(III) \[\ce{P ->[Na2Cr2O7][H2SO4] R}\]
Q and R are respectively:
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which following reagent is used to obtain alkene from alcohol?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the role of PDC from the following.
The acid, which contains both -OH and -COOR groups is ______.

Product (B) in this reaction is:
The product C in the following reaction is

Convert the following :
cumene to phenol.
Arrange the following compounds in order of decreasing acidity: