Advertisements
Advertisements
प्रश्न
Answer in brief.
Give a simple chemical test to distinguish between ethanol and ethyl bromide.
Advertisements
उत्तर
i. Ethanol on reaction with a very strong base like alkali metal Na or K gives sodium or potassium ethoxide with the liberation of hydrogen gas.
\[\ce{2CH3CH2 - OH + 2Na -> 2CH3CH2O^-Na+ + H2_{(g)}↑}\]
ii. Ethyl bromide on reaction with sodium metal does not liberate hydrogen gas.
\[\ce{2CH3CH2Br + 2Na -> CH3CH2 - CH2CH3 + 2NaBr}\]
APPEARS IN
संबंधित प्रश्न
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Write the name of major product when anisole reacts HI at 398 K
Write the reaction involved in the preparation of phenol from benzene sulphonic acid.
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
Write the reactions for the preparation of carbolic acid from aniline.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
Which of the following compounds does not react with bromine in alkaline medium?
Bromination of phenol, will NOT give:
Which of following elements does not react with hot concentrated sulphuric acid?
In Dow process, chlorobenzene is converted into phenol by ____________.
The most resistant alcohol towards oxidation reaction is:
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
(I)

(II) \[\ce{P + Zn ->[\Delta] Q}\]
(III) \[\ce{P ->[Na2Cr2O7][H2SO4] R}\]
Q and R are respectively:
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which of the following alcohols is least soluble in water?
Identify the product obtained when phenol is treated with bromine water?
Identify the compound having highest boiling point from the following?
The acid, which contains both -OH and -COOR groups is ______.
The chemical test that distinguish between benzoic acid and phenol is ______.
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Propene can be converted into propan-1-ol by:
What is the commercial method of preparation of phenol?
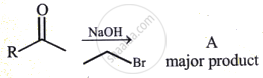
The structure of A in the given reaction is:
Which of the following is trihydric phenol?
