Advertisements
Advertisements
प्रश्न
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
Advertisements
उत्तर
i. The ether (A) with molecular formula C5H12O is
\[\begin{array}{cc}
\ce{H3C - CH2 - O - HC -CH3}\\
\phantom{..............}|\\
\phantom{................}\ce{CH3}\\
\end{array}\]
ii. Reacts with hot HI to produce two alkyl halides as follows:

iii. Oxidation of (B) gives acid
\[\ce{CH3 - CH2 - OH ->[{[O]}] \underset{(D)}{CH3COOH}}\]
iv. Oxidation of (C) gives ketone
\[\begin{array}{cc}
\ce{CH3 - CH - OH ->[{[O]}] CH3 - C = O}\\
\phantom{...}|\phantom{....................}|\\
\phantom{....}\ce{CH3}\phantom{...........}\ce{(E)}\phantom{...}\ce{CH3}\\
\end{array}\]
Hence, structural formulae of compounds (A) to (E) are
(A) (2-Ethoxypropane)
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH3}\\
\phantom{.....................}|\phantom{...........}\\ \phantom{.......................}\ce{CH3\phantom{...........}}\\
\end{array}\]
(B) (Ethanol)
CH3CH2–OH
(c) (Propan-2-ol)
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
\phantom{..}|\\
\phantom{....}\ce{OH}\\
\end{array}\]
(D) (Ethanoic acid)
CH3COOH
(E) (Propanone)
\[\begin{array}{cc}
\ce{CH3 - C = O}\\
\phantom{...}|\phantom{.}\\
\phantom{....}\ce{CH3}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Answer in one sentence/ word.
Write the IUPAC name of alcohol having molecular formula C4H10O which is resistant towards oxidation.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Write IUPAC name of crotonyl alcohol.
Write chemical equation of acetyl chloride with phenol
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
Which of the following compounds does not react with bromine in alkaline medium?
In phenols, −OH group is attached to ___________ hybridised carbon.
Phenol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
The number of isomeric alcohols possible with the formula C4H10O is ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
Identify 'A' and 'B' in the following series of reactions.
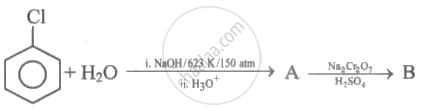
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
+I effect of alkyl groups in alcohols increases the stability of ____________.
Which of the following conversion explains the acidic nature of alcohols?
Which following reagent is used to obtain alkene from alcohol?
The number of σ bonds in carbolic acid is ______.
Identify product B in the following conversion?
\[\ce{Phenol ->[NaOH] A ->[CH3I] B}\]
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
The compound which reacts fastest with Lucas reagent at room temperature is ______.
Name the catalyst used in commercial method of preparation of phenol.
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
The order of reactivity of hydrogen halides with ether is as follows.
What is the action of nitrous acid on aniline?
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
Propene can be converted into propan-1-ol by:
